Corrosion Inhibitors
Paper
01194
ENVIRONMENTALLY FRIENDLY VOLATILE
Corrosion Inhibitors
Christophe Chandler
Cortec Corporation
4119 White Bear Parkway
Saint Paul, Minnesota 55110
U.S.A
Abstract
Volatile corrosion inhibitors (VCIs) were originally developed to protect boilers and piping systems of ships to be mothballed. Their effectiveness and ease of application attracted early users. Over the years, the field of usage has increased to cover electronics, packaging, process industries, reinforced concrete, coatings, and metalworking fluids.
Having clean parts is one of the main reasons of the ever-widening acceptance of VCIs. For example, applicators and end-users do not have to be concerned about removal and disposal as they would with petroleum-based products.
A new class of VCIs has been developed in harmony with the concern for the environment. While these new chemicals offer excellent protection to metal surfaces, they have a very low impact on the environment.
Corrosion protection and effect on the environment of several of these new VCIs were studied. Performance in typical applications was also investigated. These results are presented in this paper.
Introduction
One morning in 1943, a scientist opened a desiccator that contained some experimental nitrite-based compounds used for corrosion inhibition of the interior of gasoline pipelines1-4. As he lifted the lid of the desiccator he noticed white needle-like crystals that had formed on the inside of the cover. It turned out that these crystals were the pure amine nitrite compound placed at the bottom of the container. The chemical compound had migrated to the lid via sublimation. The scientist was quick to realize that combining the corrosion—inhibiting properties of the nitrite compound with its volatility could lead to new ways of alleviating the oxidation of metals5-11.
Like aqueous corrosion, atmospheric corrosion of metals has also been the object of numerous efforts to mitigate it. The individual and joint actions of humidity, oxygen and various corrosive gases lead to severe damage if left unchecked. As there is no limit of oxygen availability in the open atmosphere, atmospheric corrosion of metals is mainly controlled by humidity. However, aggressive gases such as hydrogen sulfide, sulfur dioxide, and ions such as chlorides control the metal attack in industrial sites and marine environments.
There are several ways of preventing atmospheric corrosion. While methods such as the use of an inert atmosphere, dehumidification, or coatings are fine in some applications, they may not be appropriate in other situations due to cost or practicability.
Because of their volatility at ambient temperature, VCI compounds can readily reach inaccessible crevices in metallic structures. Protective vapors disseminate within an enclosed space until equilibrium is reached. This equilibrium is determined by the partial vapor pressure of the VCI compound.
The US Navy was one of the very first commercial users of VCI technology. Following the end of the Second World War, several warships had to be mothballed. The areas of concern were the boilers and piping systems. The US Navy required a method that would be cost effective and easy to carry out. Responding to these requirements, VCI technology was selected and is still used by the US Navy to this day. Its range of applications has widened to include on-board electronics and other corrosion sensitive equipment12,13.
Besides its ease of use and cost effectiveness, VCI technology offers several other advantages14. One of them is cleanliness. Parts can readily be handled at any time during the protection stage or thereafter. There is no superficial film to be removed or disposed of.
Another benefit is safety. Beyond the fact VCI use does not produce unsafe work conditions, most VCI compounds have a low level of toxicity. A new generation of VCI compounds was developed as concerns grew over safety issues with the original VCIs such as dicyclohexylammonium nitrite15.
Over the years, the application field of VCI technology has increased to cover electronics16,17, packaging18,19, process industries20,21, coatings22, and metalworking fluids23.
Corrosion Testing
There are several methods to measure the effectiveness of vapor phase corrosion inhibitors. One method that is used quite widely is the Vapor Inhibiting Ability (VIA) test method24. It was developed to rapidly assess the protection offered by VCI compounds. The tested products can be powders, liquids or packaging products such as papers or plastic films. Its ease of use made this method attractive and it is still used by numerous laboratories dealing with VCI technology. The test consists of placing a freshly polished carbon steel specimen in a 1-liter glass jar. The jar contains a measured amount of water blended with glycerin to control the relative humidity. A control sample is made of a jar containing only a steel specimen, while a test sample comprises of a jar with a steel specimen along with the VCI source (powder, liquid or packaging product). The VCI source never comes in contact with the metal specimen. After a conditioning period during which the VCI vapors are allowed to migrate from the source to the metallic specimens, the jars are placed in an oven set at an elevated temperature for a few hours. Once this time elapsed, the jars are placed at ambient temperature. The metal specimen is rapidly cooled leading to condensation due to the humid atmosphere. Effective VCI compounds provide protection in this environment, while the control specimen heavily corrodes.
Environmental Aspects
Due to stricter environmental regulations, cost effectiveness aspects and other factors, car manufacturers are switching to VCI technology. North American and European automotive concerns have established guidelines to change the way of protecting parts in transit or storage. Extended testing was carried out to validate the use of VCI technology.
In addition, the discharge of chemicals used as corrosion inhibitors in a marine environment such as the North Sea oilfields has been under an ever-increasing scrutiny due the potential impact on aquatic life. Once the chemicals are disposed of in the sea, there are concerns that some of them will persist and will have a detrimental effect on the environment. These compounds may be toxic to marine life, have a low level of biodegradability or may bioaccumulate in living organisms. The Paris Commission (PARCOM) developed a protocol consisting of three tests: bioaccumulation, biodegradation and toxicity.
Bioaccumulation. Bioaccumulation of substances within aquatic organisms can lead to toxic effects over long periods of time even when actual water concentrations are low. The potential for bioaccumulation is determined by measuring the n-octanol/water partition coefficient of a specific chemical compound. The partition coefficient is the ratio of the equilibrium concentrations of a dissolved substance in a two-phase system of two largely immiscible solvents. In this case, it is defined as:
Pow concentration in n-octanol (1)
The n-octanol/water partition coefficient may be determined using high-performance liquid chromatography (HPLC)25-28.
According to the Organization for Economic Co-Operation and Development (OECD) Guideline 11729, the log Pow must be lower than 3.
Biodegradation. Biodegradation is a measure of the length of time over which a substance will remain in the environment. The OECD 306 test guideline30 is primarily used for biodegradation in marine environments. Chemical compounds are subjected to a 28-day Biochemical Oxygen Demand (BOD-28) test. The start of degradation occurs when 10% of the substance has been degraded. In order to be rapidly degradable, at least 60% degradation of the substance must be attained within 10 days of the start of degradation.
The absence of rapid degradation in the environment can mean that a chemical compound in the water has the potential to exert toxicity over a wide temporal and spatial range.
Aquatic Toxicity. Toxicity testing is run on organisms related to different levels of the food chain. This includes primary producers such as algae (Skeletonema costatum), consumers such as fish and crustaceans (Acartia tonsa), and sediment reworkers such as seabed worms (Corophium volutator).
The toxicity is usually assessed by determining an algae species 72 or 96 hour EC50, a crustacea species 48 hour EC50 and a sediment reworker 240 hour LC50.
EC50 is the effective concentration of a chemical substance necessary to negatively affect 50% of the aquatic organism population. LC50 is the effective concentration of a chemical compound required to kill 50% of the population.
EXPERIMENTAL
A volatile corrosion inhibitor identified as VCI A was used in the corrosion and toxicity tests. VCI A is made of amine carboxylates. The lower end of its melting point range is 188ºC.
Corrosion Testing
An assembly pictured in Figure 1 was used for corrosion testing. Carbon steel UNS G1018()(Fed. Steel Spec QQ-S-698) plugs (1.6 cm diameter, 1.3 cm long) were polished with a 240-grit silicon carbide abrasive. The abraded surface was then polished with a No. 400 aluminum oxide paper at 90º to the previous abraded marks. The plugs were cleaned with methanol, allowed to air-dry and then placed in a desiccator.
Ten milliliters of a synthetic glycerin-water solution having a specific gravity of 1.075 at 24ºC to create a 90% Relative Humidity atmosphere was introduced into the bottom of the test assembly.
Five hundredths of a gram of VCI were placed in a dish. The dish was then placed on the bottom of the jar. A lid was placed on the jar, tightened and the junction of the glass and lid sealed with tape.
The whole assembly was then exposed to a temperature of 24ºC for 20 hours. Then cold water at a temperature of 4ºC below the ambient was added to the aluminum tubes until full. After 3 hours the water was removed from the tubes. The steel plugs were evaluated for signs of corrosion.
In this test method, a visible change in the surface finish such as pitting or etching is considered as corrosion. Stain alone does not constitute corrosion.
Toxicity Testing
Bioaccumulation. The partition coefficient Pow was determined according to OECD-Guideline test number 117 (Partition coefficient (n-octanol/water), High performance Liquid Chromatography (HPLC) method).
Biodegradability. The biodegradability of VCI A was determined according to the OECD-Guideline test number 306.
Aquatic toxicity. The following species were used to determine the aquatic toxicity of VCI A.
Algae test. For the toxicity of VCI A to algae, the ISO/DIS 10253 (4th working draft water quality-Marine algae growth test with Skeletonema costatum and Phaoedactylum tricornutum) test method was used.
Crustacean test. ISO/DIS 14669-1997 (Water quality-Determination of acute lethal toxicity to marine copepods) was used to study the toxicity of VCI A to consumer species.
Sediment reworker. The effect of VCI A on sediment reworkers was determined using the ASTM E1367-90 test method (Standard Guide for conducting 240 hour static sediment toxicity tests with marine and estuary sediments).
Results
Corrosion Testing
The results for the Vapor Inhibiting Ability test are shown in Table 1.
The control plug had heavy corrosion, while plugs placed in the presence of VCI A had no signs of corrosion. The test samples were run in triplicate. The appearance of the plugs at the completion of the test is displayed in Figure 2.
Toxicity Testing
Bioaccumulation. The measured value of the logarithm of the partition coefficient, log Pow, is reported in Table 2.
Its value is below zero indicating the unlikeness of bioaccumulation.
Biodegradability. The 28-day Biochemical Oxygen Demand value for VCI A is reported in Figure 3.
VCI A started to degrade quite rapidly. It was 10% decomposed in less than two days. At Day 7, it was 76% degraded. It was fully decomposed at Day 27. Ten days after the start of the degradation, the level of biodegradation was above 60%, indicating that VCI A could be classified as a rapidly degradable substance.
VCI A was compared to sodium benzoate, which is used as a corrosion inhibitor. It is also a carboxylic salt. VCI A fully biodegraded as its BOD-28 value reached 100%, while the BOD value for sodium benzoate leveled off at 80%.
Aquatic toxicity. The aquatic toxicity test results are reported in Tables 3, 4, and 5.
Discussion
As the tests demonstrated, VCI A provides protection against corrosion while being innocuous to the environment.
The VIA test is a method of determining the effectiveness of a volatile corrosion inhibitor. Following the conditioning period, VCI A was able to protect a steel specimen in a moisture condensing environment. The VCI source never came in contact with the steel plug, proving that VCI A reached the metal surface via sublimation.
As indicated by the value of the partition coefficient, Pow, VCI A has a very low potential to bioaccumulate in aquatic organisms. This means that VCI A would very unlikely have toxic effects on aquatic life over long time spans. This is an important benefit as there are applications where corrosion inhibitors are discharged in marine environments.
Another factor that indicates the innocuity of VCI A to the environment is its quick biodegradability. VCI A is fully decomposed in less than 28 days. Substances that rapidly biodegrade can be quickly removed from the environment. The impact of VCI A on the marine environment is very limited.
OECD 306 refers to several levels of acute and chronic toxicities. In the case of algae or other aquatic plants, the upper limit for acute toxicity measured as 72 EC50 is 100 mg/l. According to the test results, the 72 EC50 for VCIA is 240 mg/l. This indicates that VCI A is not classified as an acute toxicant per the criteria defined in the test guideline.
It was reported in the Bioaccumulation section, log Pow for VCI A is less than 3. Furthermore, VCI A is a substance that rapidly biodegrades as stated in the Biodegradability section. Therefore, with a 72 EC50 above 100 mg/l, VCI A is not classified as a chronic toxicant.
Similar statements may be made in regards to the crustacean and sediment reworker test results.
In 1995 the Norwegian Pollution Control Authority (SFT) implemented the OSPAR Harmonized Offshore Chemical Notification Format (HOCNF) to document environmental properties of chemicals used in offshore applications. The HOCNF format contains data on toxicity, biodegradation and bioaccumulation of chemicals. The SFT issues discharge permits. The permits are issued to oil companies operating in the North Sea. They also require that the operator gradually and systematically replace chemicals that have detrimental effects on the environment with environmentally friendly products.
A large Norwegian oil and gas producing company operates several offshore and onshore installations in the North Sea. In the past, a gas line installation would be hydrotested and protected with an oil-water emulsion. As the regulations about disposal came in place, this oil company looked for other ways of protecting its equipment. After several evaluations, it selected VCI technology as it proved to be a cost effective and environmentally friendly method. The use of an oil-water emulsion lead to disposal cost as this product could not safely be disposed in the sea. In addition, for gas pipelines, an absolutely clean surface is necessary for proper use of the installation. The oil-water emulsion would leave residues after hydrotesting. Subsequent cleaning of the surface was required following the test operation.
VCI products have now been used for offshore and onshore applications for several years. VCI A is used during hydrotesting and for preservation of internal surfaces of pipes and vessels. This is carried out on large pipe systems on offshore platforms or smaller systems on refineries or on onshore oil and gas receiving stations.
It follows the same strict discharge requirements. After hydrotesting the aqueous solution of VCI A powder is sent to a storage tank and used again or simply discharged into the sea. The concentration used for this application is based on the desired length of protection and is usually between 1% and 3.5%.
VCI A was fogged inside the openings of recently hydrotested metering stations (Figure 4) at a ratio of 500 g per cubic meter. The final step involved capping the ends to seal the installation. The units were thus protected for extended periods of time until they were dispatched and installed at their final destination. The VCI powder was either left in place with no detrimental effect to the operations or simply flushed with water, and subsequently disposed of in the sea.
Other applications where VCI technology is now used include onshore pipeline (Figure 5) and pig launching (Figure 6) installations. Solutions of VCI A were used to hydrotest and protect these pieces of equipment. The solutions were sent to a storage tank for future use or discharged in the sea.
Conclusions
First developed for a few specific applications, volatile corrosion inhibitors are now used over a wide range of situations where atmospheric corrosion damages exposed metals. These metals can belong to steel structures, metal parts in transit or storage, or electronic devices. VCI compounds provide effective corrosion protection while offering several other benefits, such as cost effectiveness and cleanliness.
A new generation of VCI has been developed to address the toxicity concern that some of the original VCIs have. Corrosion and toxicity testing demonstrated that these new compounds not only provide excellent corrosion protection, but also have a very low toxicity level when placed in a marine environment. As a consequence, the oil and gas producing industry in the North Sea now uses VCI technology with great success.
ACKNOWLEDGEMENTS
The author acknowledges the assistance of Ole Lilland with Presserv AS, Stavenger, Norway, in providing the field work information.
References
- Vapor-Phase Inhibitors, Modern Packaging, December 1948: p. 147.
- VPI Goes to War, Modern Packaging, July 1951: pp. 92-95.
- VCI, Modern Packaging, October 1953: pp. 157-160.
- The Volatile Corrosion Inhibitors, Modern Packaging, November 1960: pp. 116-117.
- Cox, A.B. and Kuster, E.C., Corr. Prev. and Control, 1956: pp. 3,4 SIII.
- Berwick, I.D.G. and Levelton, B.H., Eng. Journal, 1954: pp. 37, 1, 128.
- Baker, H., Ind. Engng. Chem., 1954: pp. 46, 2592.
- Stroud, E.G. and Vernon, W.H.J., Appl. Chem., 1952: pp. 2, 166.
- Rhodes, C.A., Corr. Prev. and Control, 1957: pp. 4, 37.
- Rosenfel’d, I.L., Persiantseva, V.P., Polteva, M.N. and Terentiev, B., 1st European Symposium on Corrosion Inhibitors, Proc. 1st SEIC, Suppl. No. 3, 329, (Ann. Univ. Ferrara, N.S., Sez. V., 1961).
- Miksic, B.A. and Miller, R.H., 5th European Symposium on Corrosion Inhibitors, Proc. 5th SEIC, Suppl. No. 7, 217, (Ann. Univ. Ferrara, N.S., Sez. V., 1980).
- Vasanth, K.L., CORROSION/96, paper no. 233, (Denver, Colorado, NACE, 1996).
- Vasanth, K.L., Dacres, C.M., CORROSION/97, paper no. 179, (New Orleans, Louisiana, NACE, 1997).
- Miksic, B.A., CORROSION/83, paper no. 308, (Anaheim, California, NACE, 1983).
- Cole, D.E., et al., CORROSION/99, paper no. 495, (San Antonio, Texas, NACE, 1999).
- Miksic, B.A. and Martin, P.J., 6th European Symposium on Corrosion Inhibitors, Proc., pp. 941-950, (Ferrera, Italy, 1985).
- Sparrow, G.R. and Foley, J., Symposium on Corrosion Reliability Electronic Materials and Devices, The Electrochemical Society, Proceedings, (Miami, Florida 1984).
- Chang, Yew C. and Miksic, B.A., CORROSION/99, paper no. 93, (San Antonio, Texas, NACE, 1999).
- Rudman, B. and Chandler, C., EOS/ESD Symposium, Proceedings, (Orlando, Florida, 1996).
- Groysman, A., CORROSION/98, paper no. 240, (San Diego, California, NACE, 1998).
- Andrev, N.N. and Kutznetsov, Yu. I., CORROSION/98, paper no. 241, (San Diego, California, NACE, 1998).
- Gelner, L., CORROSION/98, paper no. 712, (San Diego, California, NACE, 1998).
- Sparrow, G.R. and Chandler, C., CORROSION/95, paper no. 489, (Orlando, Florida, NACE, 1995).
- Federal Test Method Standard No. 101C, Method 4031 (1980).
- Lyman, W.J., Reehl, W.F., and Rosenblatt, D.H. (ed.), Handbook of Chemical Property Estimation Methods, (McGraw-Hill, New York, 1982).
- Dunn, W.J., Block, J.H., and Pearlman, R.S. (ed.), Partition Coefficient, Determination and Estimation, (Pergamon Press, Elmsford, New York and Oxford, 1986).
- Pomona College, Medicinal Chemistry Project, Claremont, California 91711, USA, Log P Database and Med. Chem. Software (Program CLOGP-3).
- Hansch, C., and Leo, A.J., Substituent Constants for Correlation Analysis in Chemistry and Biology, (John Wiley, New York, 1979).
- OECD 317 Test Guidelines, Organization for Economic Co-Operation and Development.
- OECD 306 Test Guidelines, Organization for Economic Co-Operation and Development.
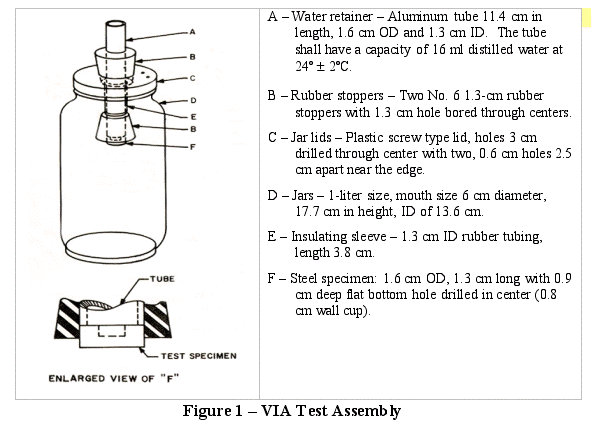
Figure 2 Ü VIA Steel Plugs
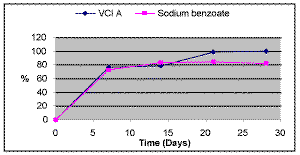
Figure 3 Ü VCI A Aerobic Biodegradability
Figure 4 Ü Metering Station
Figure 5 Ü Onshore Pipeline Installation
Figure 6 Ü Pig Launching Installation
|
Sample |
Results |
|
Control |
Fail |
|
VCI A |
Pass |
Table 1 — VIA Test Results
|
Test Method |
Result |
Limit |
|
OECD 117 log Pow |
<0 |
<3 |
Table 2 — Partition Coefficient
|
Exposure time, Hours |
Effect Concentration (mg/l) |
Limit (mg/l) |
|
|
EC50 |
72 |
240 |
³ 100 |
|
EC90 |
72 |
680 |
- |
|
No observed effect concentration |
72 |
32 |
- |
Table 3 — VCI A Toxicity to Primary Producers
|
Exposure time, Hours |
Effect Concentration (mg/l) |
Limit (mg/l) |
|
|
LC50 |
48 |
100 |
³ 100 |
|
LC90 |
48 |
220 |
- |
|
No observed effect concentration |
48 |
32 |
- |
Table 4 — VCI A Toxicity to Consumers
|
Exposure time, Hours |
Effect Concentration (mg/kg) |
Limit (mg/l) |
|
|
LC50 |
240 |
1410 |
³ 100 |
|
LC90 |
240 |
2800 |
- |
|
No observed effect concentration |
240 |
1014 |
- |
Table 5 — VCI A Toxicity to Sediment Reworkers




